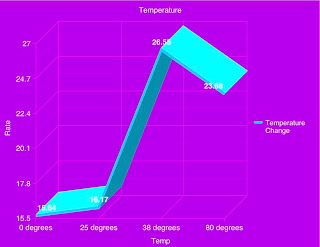
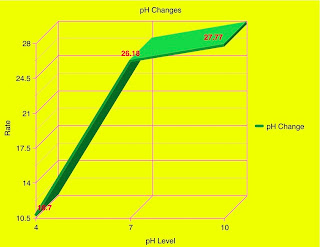
Materials: beaker, snail, elodea plant, water, Bromothymol blue, a heat lamp, and a dark room.
1)
Step 1: Take the beaker fill it to 100 ML.
Step 2: Put 20 drops of BTB into the beaker
Step 3: Stir and write down results
Step 4: Clean out beaker.
Step 5: Put 100 ML of water into beaker
Step 6: Place aquarium snail in to beaker
Step 7: Put 20 drops of BTB into the beaker
Step 8: Let sit and writed down results
Step 9: Clean out beaker
Step 10: Put 100 ML of water into beaker
Step 11: Place elodea into beaker
Step 12: Put 20 drops of BTB into beaker
Step 13: Let sit under heat lamp and write down results
Step 14: Clean out beaker
Step 15: Put 100 ML of water into beaker
Step 16: Place both the aquarium snail and elodea into beaker
Step 17: Put 20 drops of BTB into the beaker
Step 18: Let sit under heat lamp for 3 hours and write results
Step 19: Clean out beaker
Step 20: Put 100 ML of water into beaker
Step 21: Place both the aquarium snail and elodea into beaker
Step 22: Put 20 drops of BTB into the beaker
Step 23: Leave in a dark room for 3 hours
Step 24: Come back and write down results.
2)
1. Water plus bromothymol blue is blue-green because BTB is a blue-green liquid which change to a yellow color in acid and back to blue-green when returned to a neutral pH.
2. Water plus bromothymol blue plus an aquarium snail turns yellow because animals respire, then carbon dioxide in water produces carbonic acid, and then the BTB that is a blue-green liquid which changes to a yellow color in acid and back to blue-green when returned to a neutral pH.
3. Water plus bromothymol blue plus elodea, an aquarium plant, is blue-green in light because green plants photosynthesize in the light and respire all the time, then carbon dioxide water produces carbonic acid, then BTB is a blue-green liquid which changes to a yellow color in acid and back to blue-green when returned to a neutral pH.
4. Water plus bromothymol blue plus a snail plus elodea is blue-green in light and yellow when left in the dark for three hours because the animals respire and the green plants photosynthesize in the light and respire all the time that leads to sugar plus oxygen yields carbon dioxide plus water and energy because of the animal and carbon dioxide plus water yields sugar and oxygen when chlorophyll and sunlight are present that's because of the plant. Both of those make carbon dioxide in water produces carbonic acid that leads to BTB is a blue-green liquid which changes to a yellow color in acid and back to blue-green when returned to neutral pH.
I understand the lab now and that each thing is just a step to the next and the next.